Long Covid Summary Series Part 2: What causes Long Covid?
The potential root causes are as diverse and variable as its manifestations.
This is Part 2 of the Long COVID summary series. In Part 1, I discussed the challenges of determining the incidence and prevalence of Long COVID among survivors. These challenges stem from the variability in symptoms, the time since onset, individual host risk factors, inconsistencies in reporting, follow-up periods, and differing inclusion criteria across studies. In Part 2, we explore the many potential root causes that have been theorized, with some supported by some evidence.
Manifestation: As wide and variable as the hypothesized root causes
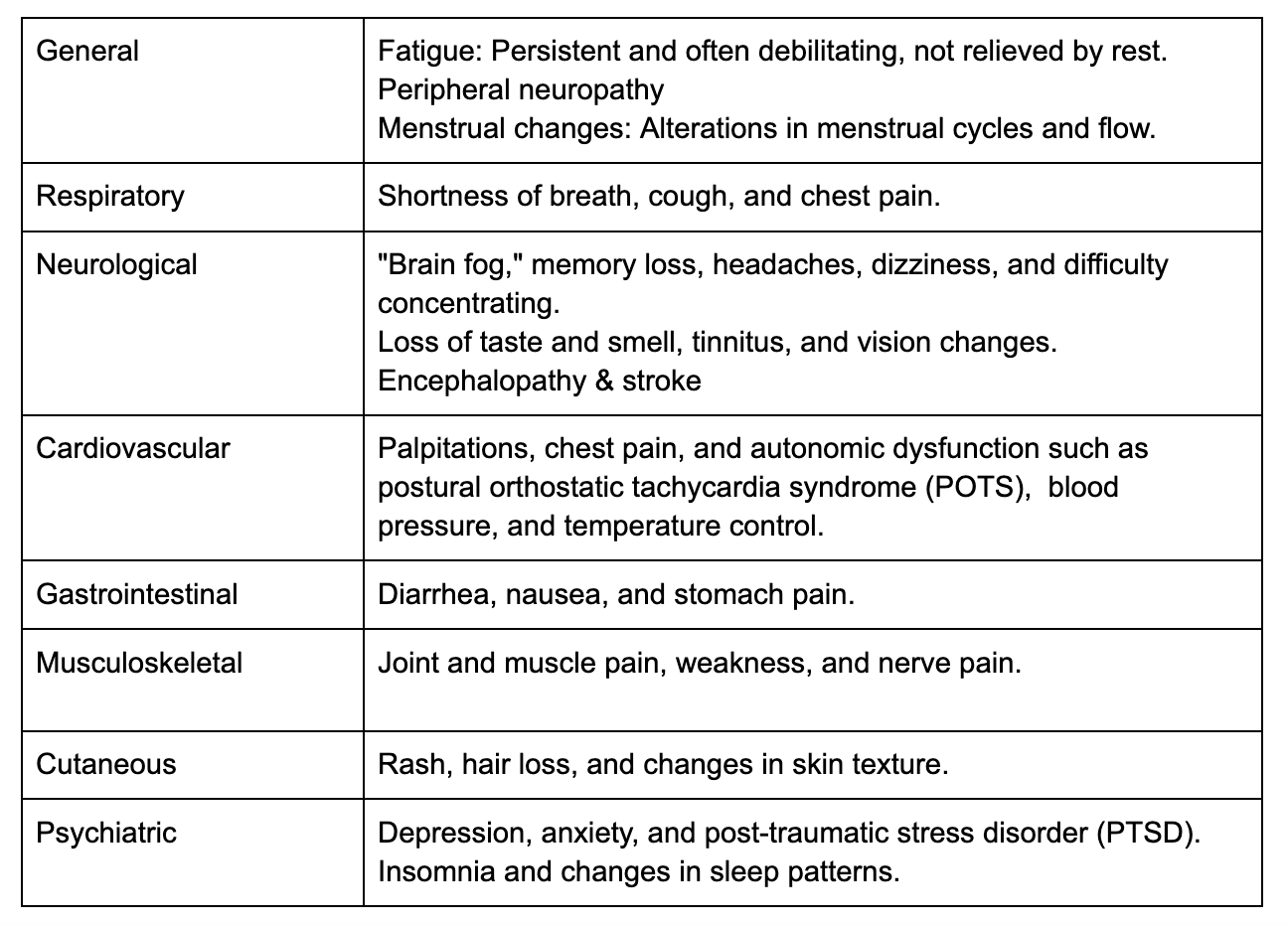
While PASC symptoms can vary from person to person and may fluctuate over time, some of the most common manifestations involve neurological, cardiovascular, pulmonary and subcutaneous systems. Some of the more widely reported symptoms relate to brain fog, fatigue and sensorial manifestations such as loss of taste and smell, but may well encompass more severe and debilitating symptoms.
A high risk of autoimmune disease has also been reported. Compared to non-COVID controls, COVID-19 patients have been found to have an increased risk of developing several autoimmune disorders, including pernicious anemia, spondyloarthritis, rheumatoid arthritis, psoriasis, pemphigoid, Graves' disease, antiphospholipid antibody syndrome, immune-mediated thrombocytopenia, multiple sclerosis, and vasculitis.
Notably, COVID-19 patients who had two doses of a COVID-19 vaccine had a decreased risk of pemphigoid, Graves' disease, antiphospholipid antibody syndrome, immune-mediated thrombocytopenia, systemic lupus erythematosus, and other autoimmune arthritis. Furthermore, the risk of COVID-19-associated autoimmune diseases varied across different age groups.
Among individuals with preexisting autoimmune conditions, COVID-19 has been found to increase the risk of developing another autoimmune disease by 23%. Patients with a more severe course of COVID-19 were at a greater risk for incident autoimmune disease, and the strongest association was with vascular autoimmune diseases.
The wide variability in symptoms runs in line with the wide range of potential root causes, which I will outline ahead.
What Causes Long Covid Symptoms?
The hypothesized root causes for Long Covid symptoms are various. Here we examine some of the evidence. There have been more updates since this was first put together, and I will leave those for a future post in the next few days.
1. Persistent Viral Reservoirs and Fragments
Several lines of evidence suggest that persistent viral reservoirs and fragments, which remain long after the infection subsides, may contribute to Post-Acute Sequelae of SARS-CoV-2 (PASC) symptoms and exacerbate the body’s inflammatory response. There are, however, many other potential root causes that have gathered evidence, such as immune dysregulation, autoimmunity, vagus nerve damage, and reactivation of dormant viruses like Epstein-Barr.
Studies have found that SARS-CoV-2 RNA can persist in various parts of the body, including different regions of the brain, for up to 230 days after symptom onset. Autopsies of 44 patients revealed that SARS-CoV-2 is widely distributed throughout the body, even in patients who died with asymptomatic or mild COVID-19.
In non-vaccinated deceased males, SARS-CoV-2 RNA was detected in the testes, which exhibited fibrosis, vascular alterations, inflammation, and cellular irregularities. Reduced sperm counts associated with SARS-CoV-2 infection may impair hormonal function and fertility in men. Furthermore, a Duke University pathology study of five recovered patients in Singapore detected SARS-CoV-2 in the colon, appendix, ileum, liver, gallbladder, and lymph nodes. The study also found evidence of residual virus in tissues six months after recovery.
2. Changes in the Immune System: Immune Dysregulation and Chronic Inflammation
Immune dysregulation has been implicated in Long COVID. Elevated functional autoantibodies (AABs), even in mild cases, have been observed to be prompted after acute SARS-CoV-2 infection, as identified amongst 177 healthcare workers in Los Angeles at least 6 months after infection. Some individuals with long COVID (LC) exhibit persistently high levels of these autoantibodies. AABs associated with systemic lupus erythematosus (SLE) and inflammatory myopathies have been seen in long COVID patients with persistent neurologic symptoms to a greater extent than COVID convalescent controls at 8 months post-infection.
Differences in autoantibody (AAB) responses between men and women, related to the presence, timing, and clustering of symptoms linked to COVID-19 infection have also been observed. In general, women showed a stronger AAB response after asymptomatic infection, while men demonstrated a wider range and higher level of AAB reactivity following infections that were reported as somewhat mildly symptomatic.
One systems immunology modeling analysis revealed that severe COVID-19 patients exhibit a significant age-related increase in autoantibody levels against 16 targets, including amyloid β peptide, β catenin, cardiolipin, claudin, enteric nerve, fibulin, insulin receptor a, and platelet glycoprotein. Principal component analysis with spectrum decomposition and hierarchical clustering based on these autoantibodies revealed an age-dependent stratification among severe COVID-19 patients.
Long Covid patients tested have shown highly activated innate immune cells and T cells, along with elevated expression of type I and type III interferons and cytokines, persisting for months post-infection. The constant release of cytokines suggests that T cells are actively dividing and responding to an antigen. Additionally, hospitalized patients exhibited a peripheral reduction in the number of T cells, and increased expression of activation and exhaustion markers, particularly in CD8+ cells. This all coincides with data from Wuhan, also showing a reduced number of total T cells.
Persistent inflammatory markers have also been detected in the brain. Elevation of CNS cytokines/chemokines was detected after and 7 weeks post-infection in mice, despite infection confined to the lungs. A chemokine called CCL11 (eotaxin-1), shown to reduce the process by which new neurons are formed in the brain, has been found to be elevated and circulating in long COVID patients who reported brain fog vs. those who did not. It is important to note the latter was driven by outliers, so other mechanisms may also be involved.
Across the olfactory mucosa in COVID-19 patients with loss of smell, SARS-CoV-2 particles and inflammation in various cell types, including olfactory sensory neurons, have been identified. In those with persistent loss of smell, the virus remained in the olfactory mucosa, indicating that prolonged anosmia might result from ongoing viral reservoirs. Infected hamsters also developed loss of smell linked to virus spread in the olfactory mucosa and CNS, along with neuroinflammation.
3. Brain-related changes
Multiple studies indicate that the breakdown of the blood-brain barrier (BBB) and the subsequent entry of serum components and cytokines into the brain is responsible for the neurological symptoms following infection. Chemokines that inhibit neurogenesis have been found at elevated levels in LC patients experiencing brain fog compared to those who do not. There is also clear evidence of microvascular damage in the brains of deceased COVID-19 patients.
Another study across 785 participants of UK Biobank found notable brain-related differences between infected and non-infected individuals (note: not LC patients), including a significant reduction in grey matter thickness, increased markers of tissue damage in areas connected to the primary olfactory cortex and decreased global brain size in SARS-CoV-2 cases. Additionally, SARS-CoV-2-infected participants exhibited a greater cognitive decline over time, on average. Microglia and astrocyte subpopulations in the brain of infected individuals have hallmark features seen in other neurodegenerative diseases like Alzheimer's, despite no molecular traces of SARS-CoV-2 in the brain. The extent or the clinical implication of this in those with Long Covid, however, has not been ascertained.
4. Organ Involvement
Even after what is considered mild-to-moderate cases, organ involvement has been documented in the national healthcare databases of the US Department of Veterans Affairs. An average of 12 months post recovery from infection, unvaccinated COVID-19 survivors higher risk of various types of cardiovascular disease, such as cerebrovascular disorders, arrhythmias, ischemic and non-ischemic heart disease, pericarditis, myocarditis, heart failure, and thromboembolic disease relative to those who were not infected and pre-pandemic data, regardless of age, race, sex or pre-existing conditions. The risks increased with disease severity.
Abnormalities in the lungs of long COVID patients with breathlessness have been identified in xenon MRIs. 30-day survivors, even those who experienced mild cases—are at increased risk for substantial declines in kidney function.
5. Microclotting
There is substantial evidence of microclotting as well. Significant fibrin amyloid micro-clots and platelet pathology were associated with Long Covid symptoms that persisted after the recovery from acute infection amongst 80 patients. Hypertension, high cholesterol levels, cardiovascular disease, and type 2 diabetes mellitus (T2DM) emerged as the most significant comorbidities.
6. Virus-reactivation
Some evidence indicates that COVID-19 may reactivate viruses that typically remain dormant after the initial infection. It is hypothesized that as COVID-19 can disrupt the immune system, it allows these previously dormant viruses to resurface. Epstein-Barr virus reactivation has been observed in individuals with Long COVID, although the data is mixed as to whether Long Covid itself can be ascribed to EBV reactivation.
7. Vagus Nerve Damage
The vagus nerve starts in the brain and extends to the body's organs, playing a crucial role in regulating heart rate, breathing, immune response, and the release of inflammatory proteins like high mobility group box 1 protein (HMGB1). This neural communication pathway is essential for maintaining the body's balance or homeostasis. If the vagus nerve and this reflex are disrupted, it can lead to an imbalance in inflammatory protein levels, causing diseases like rheumatoid arthritis, Crohn's disease, and, as evidence suggests, respiratory issues and potentially, hyperinflammation in COVID-19 patients.
There is evidence that SARS-CoV-2 infection in the vagus nerve during the acute stage may contribute to the overproduction of proinflammatory cytokines, and impair its inflammatory reflex and overall function. Another study looked at postmortem vagus nerves from COVID-19 patients and controls and found SARS-CoV-2 RNA and inflammatory cells, mainly monocytes. RNA sequencing showed a strong inflammatory response in neurons, endothelial cells, and Schwann cells, which correlated with the amount of virus present. Additionally, across a cohort of 323 patients, a decreased respiratory rate was noted in those who did not survive critical COVID-19.
The Road to Targeted Therapies
Pinning down the underlying mechanisms of Long COVID can lead to more effective treatments and management strategies, helping to alleviate the long-term suffering of millions of individuals struggling with this condition. Identifying the distinct causes, as they pertain to the different symptoms, also enables the development of targeted therapies potentially reducing healthcare costs and improving patient outcomes. A clear understanding of Long COVID's root causes can better inform public health policies and preventive measures, minimizing the incidence and impact of this condition. Finally, uncovering the causes can provide insights into other post-viral syndromes, broadening our knowledge and ability to combat various long-term effects of viral infections.